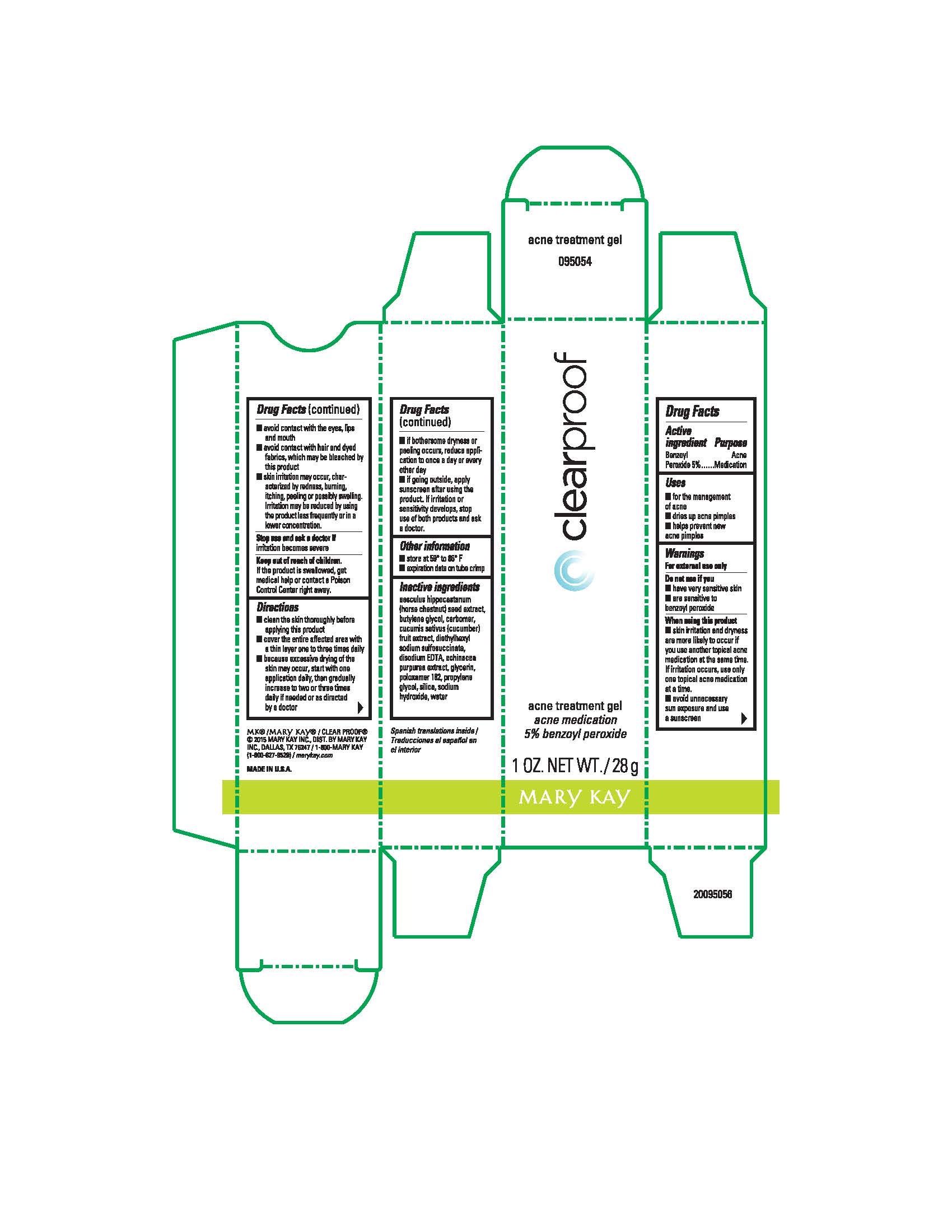
Label: CLEAR PROOF ACNE TREATMENT ACNE MEDICATION- benzoyl peroxide gel
- NDC Code(s): 51531-9071-0, 51531-9071-1
- Packager: Mary Kay Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 17, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient:
- Purpose
- Uses
-
Warnings
For external use only.
When using this product
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with the eyes, lips and mouth
- avoid contact with hair and dyed fabrics, which may be bleached by this product
-
skin irritation may occur, characterized by redness, burning, itching, peeling or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration.
-
Directions
- clean the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
- if going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use of both products and ask a doctor.
- Other information
- Inactive ingredients
- Principal Display Panel - 28 g carton
-
INGREDIENTS AND APPEARANCE
CLEAR PROOF ACNE TREATMENT ACNE MEDICATION
benzoyl peroxide gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51531-9071 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POLOXAMER 182 (UNII: JX0HIX6OAG) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) SODIUM HYDROXIDE (UNII: 55X04QC32I) DOCUSATE SODIUM (UNII: F05Q2T2JA0) EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERIN (UNII: PDC6A3C0OX) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ECHINACEA PURPUREA (UNII: QI7G114Y98) CUCUMBER (UNII: YY7C30VXJT) HORSE CHESTNUT (UNII: 3C18L6RJAZ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51531-9071-1 1 in 1 CARTON 08/16/2016 1 28 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:51531-9071-0 3 g in 1 TUBE; Type 0: Not a Combination Product 08/16/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 06/15/2012 Labeler - Mary Kay Inc. (049994452) Establishment Name Address ID/FEI Business Operations Port Jervis Laboratories Inc. 001535103 manufacture(51531-9071)