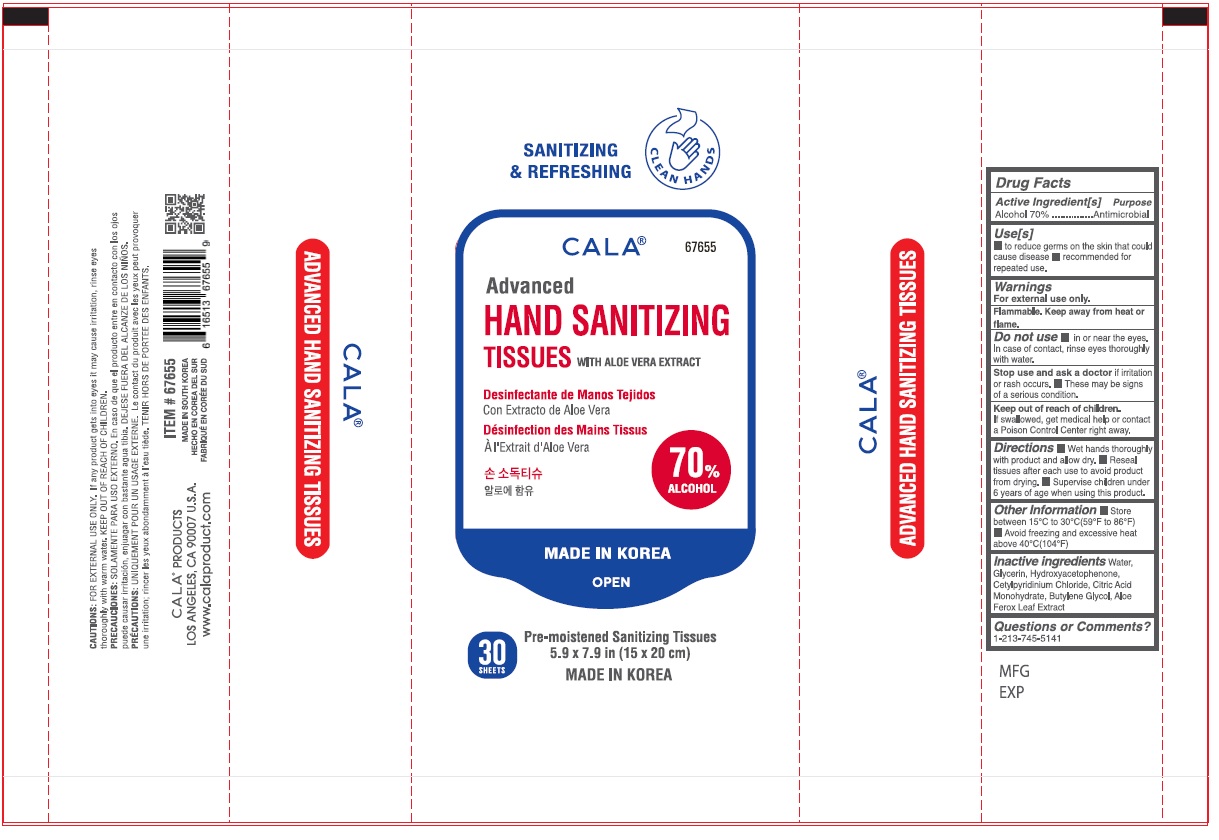
Label: CALA ADVANCED HAND SANITIZING TISSUES- alcohol swab
-
Contains inactivated NDC Code(s)
NDC Code(s): 74985-678-55, 74985-678-56 - Packager: Shin's Trading Co., Inc. Dba Cala Products
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 16, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient[s]
- Purpose
- Use[s]
- Warnings
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- Questions or Comments?
-
SPL UNCLASSIFIED SECTION
SANITIZING & REFRESHING
CLEAN HANDS
WITH ALOE VERA EXTRACT
Pre-moistened Santizing Tissues
5.9 x 7.9 in (15 x 20 cm)
MADE IN KOREA
Cleansing & Refreshing
ADVANCED HAND SANITIZING TISSUES
CAUTIONS: FOR EXTERNAL USE ONLY. If any product gets into eyes it may cause irritation, rinse eyes thoroughly with warm water. KEEP OUT OF REACH OF CHILDREN.
CALA® PRODUCTS
LOS ANGELES, CA 90007 U.S.A.
www.calaproduct.com
MADE IN SOUTH KOREA
- Packaging
-
INGREDIENTS AND APPEARANCE
CALA ADVANCED HAND SANITIZING TISSUES
alcohol swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74985-678 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) CETYLPYRIDINIUM CHLORIDE (UNII: D9OM4SK49P) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ALOE FEROX LEAF (UNII: 0D145J8EME) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74985-678-55 30 in 1 POUCH 09/11/2020 1 4.19 mL in 1 PACKET; Type 0: Not a Combination Product 2 NDC:74985-678-56 60 in 1 POUCH 09/11/2020 2 4.65 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 05/07/2020 Labeler - Shin's Trading Co., Inc. Dba Cala Products (175832021)